Shock Abortion Pill Review Ordered by Health Sec RFK Jr After Controversial Data Push

Major action has been taken regarding access to a widely used medication.
A new look at crucial health regulations is now underway.
Health Secretary Cites ‘New Data’ Prompting Review
US Health Secretary Robert F. Kennedy Jr. announced a significant directive this week.
He has ordered the Food and Drug Administration (FDA) to review the regulations surrounding the abortion pill mifepristone.

Speaking during a congressional hearing with Missouri Senator Josh Hawley, Kennedy cited “new data” as the reason for the mandated review.
He indicated that this data, while requiring further investigation, suggests at minimum, a change to the current labeling might be necessary.
“It’s alarming,” Kennedy told Senator Hawley. “Clearly, it indicates that, at very least, the label should be changed.”
This move comes as conservative organizations have amplified concerns regarding the safety profile of the medication.
Focus Turns to Recent Analysis
The “new data” referenced by the Health Secretary stems from a paper published by the Ethics and Public Policy Center (EPPC), a right-leaning think tank.
This paper, released on April 28th, suggests potentially higher complication rates associated with mifepristone than previously understood.

While this specific analysis has not undergone traditional peer review or publication in standard medical journals, it has drawn significant attention.
The authors highlight specific outcomes they believe warrant re-evaluation, such as the classification of ectopic pregnancies or the need for follow-up procedures as “serious adverse effects.”
They argue that the way certain outcomes are categorized in official data might underestimate risks.

For context, mifepristone is used in nearly two-thirds of abortions nationwide, often in combination with misoprostol.
Current medical consensus, supported by numerous studies, regards the medication as safe and effective when used according to FDA guidelines.
Calls for Reinstating Earlier Safety Measures
Based on their analysis, the EPPC paper authors propose that the FDA should consider reinstating earlier restrictions on mifepristone.
These past regulations included requirements like in-person dispensing bans on telemedicine use, and limiting the drug’s use to the first seven weeks of pregnancy.

They suggest that restoring such limits could allow for better monitoring of patient outcomes.
Some conservative groups view this potential for enhanced monitoring as a crucial step.
Such ideas align with proposals found in the Project 2025 playbook, a detailed policy guide from conservative organizations.
Project 2025 suggests ending telemedicine prescriptions for abortion pills as an “interim step” in a broader effort to ensure medication safety standards are rigorously applied.
Groups Strategize for Regulatory Change
Conservative leaders have indicated their intention to use the EPPC paper to advocate for significant changes to mifepristone access.
During a private discussion, activists reportedly referred to their strategy for influencing the FDA and lawmakers as “Rolling Thunder.”

Organizations involved reportedly included Americans United for Life, Live Action, Students for Life, and Susan B. Anthony Pro-Life America.
Their focus includes ending telemedicine prescriptions, seeing it as a crucial “first step” in ensuring all medication abortions occur under appropriate medical oversight.
Ryan Anderson, the EPPC president, reportedly argued that this approach aligns with the principle of states regulating medical practice within their borders, particularly concerning interstate medication delivery.
Addressing criticisms about the paper’s lack of peer review, some activists suggested traditional academic channels might be resistant or potentially biased against findings that challenge the current regulatory status of the drug.
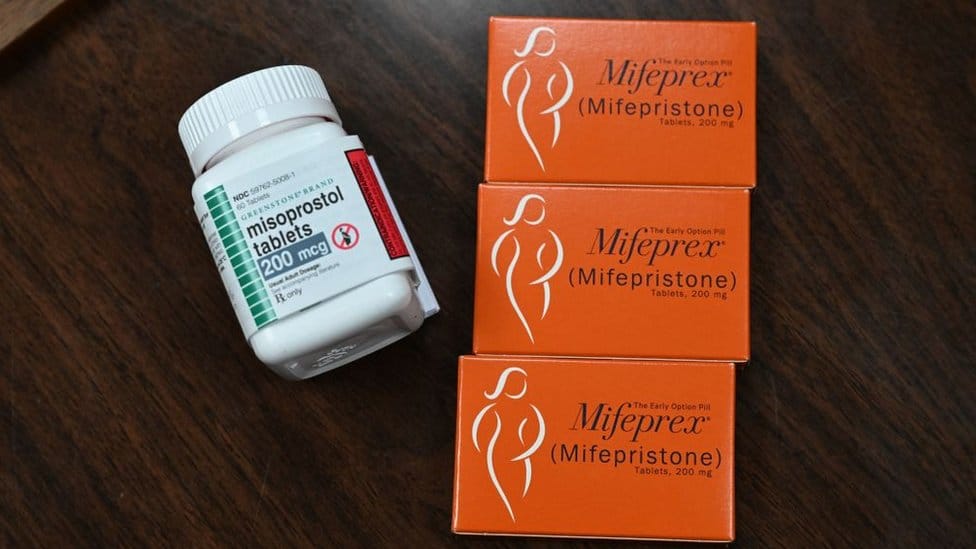
While acknowledging the paper isn’t a traditional clinical study, American Association of Pro-Life OBGYNs CEO Christina Francis reportedly stated it still warrants government action and further research into potential safety concerns.
FDA Review Officially Underway
The Health Secretary’s order formalizes a process that has been anticipated.
During his confirmation hearing in January, Kennedy mentioned that former President Trump had asked him to review mifepristone safety data.
FDA Commissioner Marty Makary also indicated in March his openness to reviewing new safety data and taking action if necessary.
Senator Hawley had specifically requested Makary review the EPPC paper shortly after its publication, and has also introduced legislation aimed at ending telemedicine access for the pill, though its passage is uncertain.
The FDA, however, has the authority to implement regulatory changes on its own, especially when presented with data raising potential safety questions.
This official review by the FDA, directed by the Health Secretary, is the direct result of these converging factors – the emergence of the new data and the advocacy efforts highlighting its implications.
It signifies a serious evaluation of the current regulatory framework for mifepristone based on the concerns raised by the EPPC analysis and supporting groups.
As this review proceeds, all eyes will be on the FDA’s findings and potential actions, which could significantly impact access to medication abortion across the United States.